health
May 19, 2023 | 2:25 PM
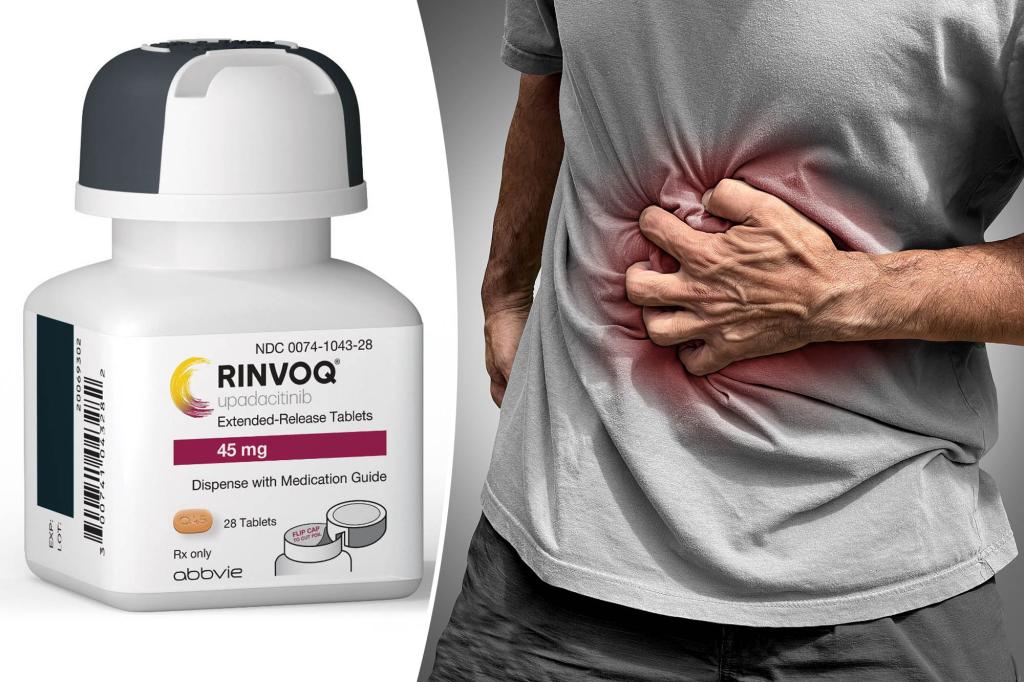
Rinvouk is the first oral medication approved by the Food and Drug Administration for moderate to severe Crohn’s disease (chronic bowel disease).
There is no cure for Crohn’s disease, and currently available drug treatments offer only some remission and relief of symptoms such as abdominal pain, diarrhea, fatigue, weight loss, bloody stools, and malnutrition. According to the Mayo Clinic.
Rinvoke (generic name: upadacitinib) FDA approved Thursday completed two clinical trials involving 857 Crohn’s disease patients.
“Within a few weeks, I started feeling better,” says Danielle, one of the Crohn’s study participants. said in a statement. “My symptoms have lessened, I have less cramping, my stools are firmer, and I have stopped bleeding. When the bleeding has stopped, I have more energy.”
Patients were randomly grouped to receive 45 mg of Rinvoque daily or placebo for 12 weeks.
Patients receiving Rinvoq were shown to have significantly less intestinal inflammation compared to the placebo group after a 3-month period.
In another study, 343 patients using low-dose Rinvouk as long-term maintenance therapy also had reduced inflammation after one year of use.
Rinvok, Made by AbbVie The North Chicago, Illinois drug belongs to a class of drugs known as Janus kinase inhibitors, which slow down the body’s immune response and reduce inflammation.
Rinvoq is already FDA-approved for the treatment of six other immune-related diseases, including rheumatoid arthritis, the skin disease atopic dermatitis, and the gastrointestinal disease ulcerative colitis.
With Rinvoq’s seventh FDA approval, the drug has been a huge success worldwide. AbbVie also manufacturesHis best-selling drug Humira (adalimumab) is a type of immunosuppressive drug known as a TNF inhibitor.

With Humira’s patent expiring in the next few years, Rinvoke is expected to increase its position as the company’s main source of revenue.
However, in recent years, the FDA has stepped up its scrutiny of all Janus kinase inhibitors following reports of serious heart-related side effects and cancer risks. According to Fierce Pharma.
As a result of these safety concerns, JAK inhibitors, including Rinvoq, are currently only approved for patients who are intolerant or do not respond well to TNF inhibitors such as Humira who have tried them.
Crohn’s disease can run in families, but doctors still don’t know what causes Crohn’s disease. In addition to immunosuppressants, treatment for this condition may include anti-inflammatory drugs such as corticosteroids, antibiotics, nutritional therapy, and, in moderate or severe cases, surgery.
load more…
{{#isDisplay}}
{{/isDisplay}}{{#isAniviewVideo}}
{{/isAniviewVideo}}{{#isSRVideo}}
{{/isSR video}}