That may have been the case with Frostbite.
The U.S. Food and Drug Administration (FDA) first medicine to treat severe frostbite, authorities announced this week.
The drug iloprost (trade name Allmin) is intended to reduce the risk of finger and foot amputation due to: dangerously cold limb.
Need a “winter reset”? Experts share the benefits of slowing down during the cold season
Iloprost was originally approved to treat pulmonary arterial hypertension, a condition in which high blood pressure affects the pulmonary arteries. lungs and heart.
“This approval provides patients with the first-ever treatment option for severe frostbite,” Norman Stockbridge, M.D., director of the Division of Cardiology and Nephrology in the FDA’s Center for Drug Evaluation and Research, said in a press release. .
Frostbite occurs when cold temperatures freeze parts of your body, primarily your fingers, toes, nose, cheeks, and jaw. (St. Petersburg)
“This new option gives doctors a tool to help prevent life-changing amputations of frostbitten fingers and toes.”
According to the Centers for Disease Control and Prevention (CDC), frostbite occurs when parts of the body, primarily fingers, toes, nose, cheeks, and chin, freeze due to low temperatures.
Ask your doctor: “How should I take care of my skin during the winter?”
Early symptoms include numbness, tingling, and color changes, says Dr. Mark Siegel, clinical professor of medicine. New York University Langone Medical Center a Fox News medical contributor told Fox News Digital.
“Severe frostbite can cause the skin to turn white or blue, followed by fluid-filled blisters,” said Siegel, who was not involved in the drug’s development.
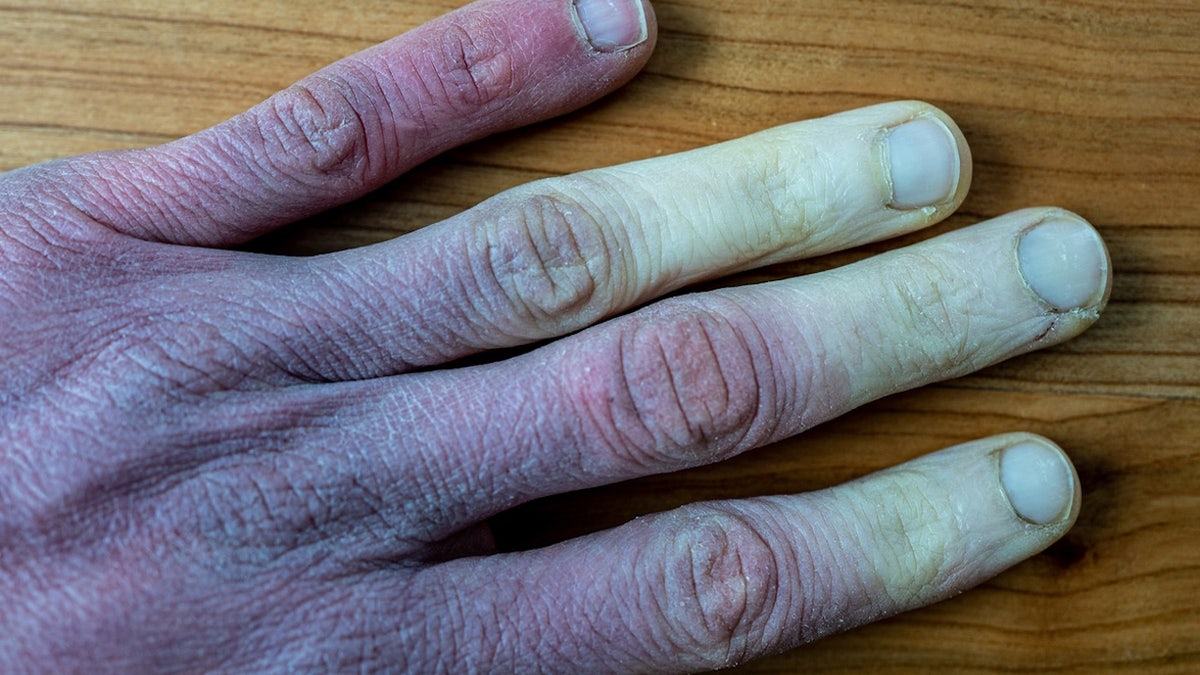
Early symptoms of frostbite include numbness, tingling, and color change. (St. Petersburg)
The CDC said prolonged frostbite can lead to permanent damage and amputation.
Aurlumyn is intended for cases of severe frostbite, where both the skin and underlying tissue have frozen and blood flow has stopped.
Why your pain increases in cold weather — and what you can do about it
“Iloprost, the active ingredient in Aurumin, is a vasodilator, a drug that opens blood vessels and prevents blood clotting,” the FDA release states.
FDA approval follows randomized results. clinical trial They included 47 adults suffering from severe frostbite, who were divided into three groups.


The CDC said prolonged frostbite can lead to permanent damage and amputation. (I stock)
One group received iloprost intravenously for 6 hours daily for up to 8 days.
Another group received a combination of iloprost with other drugs not approved for frostbite.
A third group received other drugs that did not include iloprost.
Snow shoveling safety tips to prevent injuries and heart attacks: ‘Very strenuous work’
Seven days after the initial frostbite, each participant underwent a bone scan to determine whether finger or toe amputation was necessary.
No patients receiving iloprost required amputation, compared with 19% of patients receiving iloprost in combination with other drugs and 60% of patients receiving other drugs alone.


A sign outside the Food and Drug Administration (FDA) headquarters in White Oak, Maryland, on August 29, 2020. (Reuters/Andrew Kelly/File Photo)
“This is a very important approval,” Siegel told Fox News Digital.
“Severe frostbite can cause blood clotting and lead to amputation. The active ingredient in this new drug, Ilopost, is a vasodilator that maintains blood flow to areas of severe frostbite, and a small study has prevented the need for amputation in all cases.”
Click here to sign up for our health newsletter
Allmin, developed by Maryland-based Eikos Sciences, has been found to cause some side effects, the FDA said.
These include palpitations, increased heart rate, nausea, headache, facial flushing, vomiting, dizziness, and low blood pressure.
CLICK HERE TO GET THE FOX NEWS APP
As such, Siegel said, the drug can only be administered. by a doctor.
Fox News Digital has reached out to Eicos Sciences Inc and the FDA for additional comment.
For more health articles, visit: www.foxnews.com/health.

